1869 Alizarin 1890 Indigo 1901 Indanthrene 1913 Ammonia synthesis
BASF

In 1859, William Henry Perkin, an Englishman who was only eighteen years old at the time, attempted to produce quinine synthetically. In one of his experiments, he treated aniline with the strong oxidizing agent potassium dichromate. This unexpectedly produced a dye: mauveine, also known as aniline purple or Perkin violet. Perkin realized that not only mauveine but also many other dyes could be made from aniline.
Aniline, like many other chemical compounds, is contained in the residues produced during the manufacture of illuminating gas and coke from hard coal: coal tar. Aniline and many other substances from coal tar that are still important for the chemical industry today were isolated and studied by the German chemist Friedlieb Ferdinand Runge as early as the 1930s.
Friedrich Engelhorn was born in Mannheim in 1821, the son of an innkeeper. After attending the lyceum, he completed an apprenticeship as a goldsmith and passed the master craftsman's examination in 1846.
During the Baden Revolution of 1848, Friedrich Engelhorn, as commander of the Mannheim Citizens' Militia, achieves a peaceful surrender of the city to the Prussian army, thus preventing the destruction of Mannheim.
However, his goldsmith's workshop, which was initially operated in his parents' house and later in Square C 4.6, ran into economic difficulties during the revolutionary years and Engelhorn looked for another field of activity. Together with two partners, he founded a gasworks, which started production in 1849.



Engelhorn quickly recognized the opportunities offered by the coal tar produced during gas production. Together with the chemists Carl Clemm and Nepomuk Spreng and the merchant Otto Dyckerhoff, he founded an aniline dye factory in the Jungbusch district of Mannheim on June 19, 1861 and began to produce aniline. The chemicals needed to produce aniline were initially supplied by the "Verein Chemischer Fabriken" in Mannheim.


However, as it was more economical to have the entire manufacturing process from raw material to end product in one hand, Engelhorn sought to merge with the supplier, but this was rejected by its shareholders. Engelhorn decided to produce the necessary raw materials itself and founded the Badische Anilin- und Soda-Fabrik on April 6, 1865.
But the previous production facility in Jungbusch no longer offered sufficient space. Engelhorn therefore wanted to buy a plot of land in Mannheim. Because the "Verein Chemischer Fabriken" (Chemical Factories Association) was also interested in the site, Mannheim's citizens' committee decided to put it up for public auction in order to achieve the highest possible price.
Engelhorn immediately acquired another plot of land, not in Mannheim but in Ludwigshafen. Incidentally, no one showed up at the public auction held two weeks later.
Construction of the factory on the other side of the Rhine began as early as May 1865.


Alizarin
On November 1, 1868, the renowned chemist Heinrich Caro was hired as co-director and head of research. He had studied in Berlin and then specialized in color chemistry in Manchester and London. Initially, he set up his own testing facility in the old Mannheim factory.
In May 1869, BASF acquired from Carl Graebe and Carl Liebermann, a student of Robert Bunsen, who taught in Heidelberg, the rights to the first process for the synthetic production of a natural red dye, "alizarin," which had already been extracted from madder root in ancient times.
However, Graebe and Liebermann's bromination process proved too expensive for alizarin production on an industrial scale. A coincidence helps in the search for a technically viable manufacturing process.
When Caro heats anthraquinone in a dish but briefly turns his attention to another task, he finds the laboratory smoky when he returns. A substance has formed in the dish that can be fused with caustic potash under the action of hot sulfuric acid to form alizarin.
On June 25, 1869, the English patent was granted for the inexpensive sulfonation process needed for large-scale production. Just one day later, William Henry Perkin also wanted to patent an identical process. Friedrich Engelhorn avoids a lengthy legal dispute and reaches an agreement with Perkin on joint marketing to their mutual benefit. BASF thus gains access to the world market, and Alizarin becomes BASF's first global sales success.
Just under a year later, 230 kg of Alizarin red are produced. At 270 marks per kg, it is still more than four times as expensive as natural madder red, which costs 60 marks. But thanks to a new production plant, after five years production can be increased to 100 tons, and later even to over 1,000 tons, bringing the price of a kilogram of alizarin down to 9 marks.
For over 3,000 years, the red dye was extracted from the root of the dyer's madder. Now it is no longer worth growing the plant, and the agricultural land, especially in France and the Netherlands, can now be used to grow food for the rapidly growing population.



Methylene blue
Heinrich Caro did not rest on his success with the alizarin synthesis, but continued his research. In 1875, together with his friend Professor Adolf von Baeyer, he discovered nitrosodimethylaniline and used it to develop a blue dye, methylene blue, for which he filed a patent application on December 15, 1877. This was the first German patent for a tar dye.
Like alizarin, which is not only a printing and artist's ink but is also used in histology as a dye for the microscopic examination of calcium deposits in brain tissue, methylene blue also has medical significance. When the physician Paul Ehrlich stained malaria pathogens with methylene blue during his work on the vital staining of tissue, he discovered the healing power of methylene blue against malaria in 1891. And later, Robert Koch makes the tuberculosis bacillus visible with methylene blue.

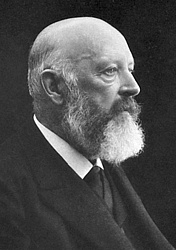

Indigo
After years of experimentation, Adolf von Baeyer succeeded in synthesizing indigo from cinnamic acid for the first time in the laboratory in 1878.
The deep blue pigment is one of the oldest and best-known natural colors and was already used in prehistoric times to dye textiles. Traces of indigo can be found in 6,000-year-old Peruvian cotton fabrics. In Egypt, mummies were wrapped in indigo-dyed mummy bands as early as 4,400 BC. Indigo was also used in early Mesopotamian cultures.
In the past, indigo was obtained from various plants containing indigo, such as the Indian indigo plant, , which gave the color its name, or the leaves of the woad, which was already planted in Europe in ancient times. Oxygen is removed from the water-insoluble dye using a solvent, usually urine. The fabric to be dyed is soaked in the yellowish-green broth for several days. Only when it dries under the influence of oxygen does the blue dye recede and bond with the fiber. Until then, the dyers have to wait with further processing. They "make blue."
However, the dye, which is laboriously produced from plants, is expensive, but the demand for blue dyeing of uniforms and work clothes is high. The synthetic production of indigo thus promises high profits. BASF was therefore very interested in Baeyer's work. When Prof. Baeyer filed his patent for indigo synthesis in 1880, BASF secured the exploitation rights.
Particularly advantageous for the dyeing process is the fact that, under suitable conditions, Baeyer's process converts the starting materials into indigo directly on the textile fiber.



As early as April 1881, BASF starts production of "Little Indigo." But Baeyer's synthesis route proves uneconomical for industrial production and the product does not find market acceptance. Production was therefore discontinued after only three years. Another, process, also leads to a dead end despite years of trials.
The immense research effort in which BASF invests 18 million gold marks over the years, as much as the company's share capital at the time, does not lead to success. In 1883, Friedrich Engelhorn leaves BASF's Board of Executive Directors and buys into the Mannheim-based pharmaceutical company C.F.Boehringer und Söhne. Carl Clemm also leaves BASF and founds the Waldhof pulp factory a year later together with his brother Adolf.


They are succeeded on BASF's Board of Executive Directors by the young chemists Carl Glaser and Heinrich Brunck. But it is not until 1890, when the Swiss professor Karl Heumann publishes a novel synthetic route for indigo using an alkaline melt of phenylglycine, that indigo research receives a new boost.
BASF is immediately interested. After all, it produces the starting products for this new process - aniline, acetic acid, chlorine and alkali - in-house. But it turns out that even with this new process, the indigo yield is too low for economical production, and further development is initially halted.
When Professor Heumann discovered that the yield could be considerably higher if other starting materials were used, work on improving the process was resumed.





After 17 years of intensive research, the synthetic dye finally appears on the market on July 10, 1897 under the name "Indigo pure B.A.S.F." and can be produced inexpensively in any quantity. For BASF, this represents a major commercial success. The "Indigo pure B.A.S.F." is in demand worldwide. Whereas the composition of natural indigo, and thus the dye content, fluctuates, synthetic indigo can be used to achieve a consistently uniform dyeing.
By the time BASF wants to stop indigo production in the 1960s, jeans are coming into fashion in Europe, especially washed-out jeans. Since only synthetic indigo achieves the desired effect, demand for it increases again. Today, indigo is produced mainly for dyeing the cotton fabric known as denim (de Nimes) for blue jeans, which thus acquire their typical appearance. When jeans lose their blue color during washing, you can then experience a blue miracle. Indigo, like the other first synthetic dyes, is not washfast. Therefore, natural dyes are still in demand.




Indanthren
But just four years after its market launch, the new indigo had competition from within the company. BASF chemist René Bohn develops "Indanthren," the first lightfast and washable blue dye.
Even as a child, René was interested in chemistry and experimented with great enthusiasm in the small laboratory set up in his father's factory. He studied chemistry at the Zurich Polytechnic from 1879 to 1882, among others under Professor Karl Heumann. At the age of 22, René Bohn was hired by BASF in the alizarin department of the company laboratory and soon attracted attention with the invention of new dyes: alizarin marron in 1885, anthracene blue in 1886, alizarin black in 1887, and carbazole yellow, alizarin green and alizarin blue-green in 1888. His best-known invention, however, was the washfast and lightfast indanthrene. To synthesize a washfast "superindigo," he used anthraquinone, which had already served as a starting material for alizarin 30 years earlier, and obtained a brilliant blue dye that he called indanthrene. On February 6, 1901, BASF filed a patent application for the new development as a "process for the preparation of a blue dye of the anthracene series.
But before indanthrene can begin its triumphal march, a number of non-technical problems have to be solved. Indanthrene is not only much more expensive than indigo, but also requires a completely different dyeing technique. This is why there are initially reservations among users, dyers and printers. Bohn therefore produces precise application instructions.
It is not only the users who have to be convinced of the new washable ink, but also the end users. In 1921, for the first time in its history, BASF launches an advertising campaign with advertisements, posters and even fashion shows. In January 1922, a trademark is registered for Indanthren.



Ammonia synthesis
The famous Justus von Liebig had already described in 1840 in his work on agricultural chemistry that plants need minerals, especially phosphorus, potassium and nitrogen, which they absorb from the soil with their roots, to form chlorophyll and plant proteins. Intensive farming, however, removes these minerals from the soil over time.
Around 1900, Germany and many other parts of Europe experience increasingly frequent crop failures and famines. Many people emigrate to America during this time.


In order to secure the supply of food for the rapidly growing population, nitrogen in particular must be added to the depleted soils.
Natural fertilization with slurry or manure, which releases ammonia through the decomposition of nitrogenous substances, is not sufficient. Ammonia is a poisonous gas at room temperature with a pungent odor that we know from its aqueous solution, ammonia solution. The nitrogen atom contained in the ammonia molecule is bonded to three hydrogen atoms, hence its formal "NH3". The N stands for nitrogen, the H for hydrogen.
In those days, saltpeter was used to supplement organic fertilizers. This nitrogen-containing salt occurs naturally, but is very expensive because it has to be brought in from overseas, e.g. from Chile. The idea of producing on an industrial scale the nitrogen salts that plants use to build up their leaf mass is an obvious one.
Nitrogen, a colorless and odorless gas, is abundant because it is the main component of the earth's atmosphere, accounting for 78% of the total. But plants cannot utilize atmospheric nitrogen directly. The nitrogen molecule N2 consists of two nitrogen atoms firmly bonded to each other. Only with a high energy input can the two atoms be torn apart.
Thunderstorm lightning, for example. The resulting high temperatures split the nitrogen atoms, which immediately combine again elsewhere, for example with the hydrogen contained in the rainwater. In this way, the nitrogen reaches the soil in a form that can be used by plants. A thunderstorm is particularly effective during the growing season in spring. It is not for nothing that an old farmer's saying goes, "Thunderstorms in May, the farmer cries juchhei!"
At the beginning of the 20th century, there is still no process to convert the inert atmospheric nitrogen into a chemical compound and thus make it usable as a fertilizer. Developing such a process is a very demanding task.
The chemist Fritz Haber, professor at the Technical University of Karlsruhe, investigates the reaction between nitrogen and hydrogen under high pressure and at high temperature. After years of experimentation, he discovered that a pressure of around 300 bar, i.e. 300 times the normal atmospheric pressure, temperatures of 400 degrees Celsius and, last but not least, suitable catalysts were required to achieve a sufficient reaction rate.
In 1908, his experimental apparatus produced the first synthetically manufactured ammonia under laboratory conditions. He registered his process for patent at and turned to BASF for its industrial-scale implementation. However, BASF sees a major problem, particularly with regard to the high pressures required. There are still no pressure vessels and pipes that can withstand such loads. In addition, hydrogen diffuses through even the thickest steel walls at high temperatures and pressures because its atoms are tiny.



But Carl Bosch, a chemist at BASF since 1899, finds a solution. He constructs a double-walled tube from new types of steel. The inside of the tube consists of soft, low-carbon iron, which is barely penetrated by hydrogen, and a jacket of solid steel. The hydrogen still diffusing through the inner tube can escape through holes in the outer tube.
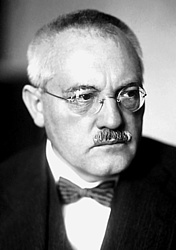


After extensive research, Alwin Mitasch, head of the ammonia laboratory at BASF and a close associate of Carl Bosch, develops a suitable catalyst made of iron with oxide components of aluminum, calcium and potassium.
The first high-pressure-resistant and thus operationally safe reactor in the history of process engineering was thus invented, and after five years of research and development Haber's laboratory result was made usable for large-scale production.
The ammonia synthesis process, named the Haber-Bosch process after its developers, is the first to succeed in combining atmospheric nitrogen with hydrogen in huge high-pressure reactors. The resulting ammonia is liquefied and separated by cooling the gas mixture. In 1913, mineral fertilizer production began at the first synthesis plant in Oppau near Ludwigshafen. By 1914, more than 7,000 metric tons of low-cost percolating fertilizer are available.
Starting in 1914, Bosch conducted field trials at BASF's own Limburgerhof Agricultural Research Station to determine the optimum quantities, application times, and distribution of fertilizer, so that the fertilizer could also provide farmers with optimum instructions for use. The farmers were introduced to the new artificial fertilizer through amusing advertising.




But then the First World War breaks out and ammonia becomes a raw material vital to the war effort, because the nitric acid extracted from it is a starting product for explosives.
Unfortunately, Fritz Haber plays an inglorious role in the First World War. He not only researches the development of gas warfare agents, but even personally supervises the first large-scale use of poison gas in history in Ypres in 1915. His chlorine gas killed around 1500 French soldiers and seriously wounded many more. Haber's wife Clara Immerwahr, the first female chemist to earn a doctorate in Germany, detested his work as a "perversion of science" and shot herself with Haber's service weapon in the garden of the Berlin service villa.



Nevertheless, Fritz Haber is awarded the 1918 Nobel Prize in Chemistry for the development of ammonia synthesis. Carl Bosch is not awarded the Nobel Prize in Chemistry for his work on large-scale ammonia synthesis until 1931, together with Friedrich Bergius, a student of Fritz Haber.


After the end of the war, ammonia and nitric acid production grows steadily. Although the risks of production and storage are well known, a catastrophe occurs on September 21, 1921, claiming the lives of 561 people when a huge explosion rocks Oppau, destroying most of it. Even in Heidelberg, 25 km away, the blast wave covers house roofs. To this day, it is the biggest chemical disaster in BASF's history.
More than three-quarters of the total production of nitrogen bound in the form of ammonia is still processed into mineral fertilizers. The rest is used in the manufacture of many other products, such as synthetic fibers and even baking powder. Car tires also contain ammonia. Nitrogen compounds such as hydrazine and nitric acid can be used to produce rocket fuel. In pharmaceuticals, the sulfonamides produced from ammonia are used for medicines such as antibiotics.





